Quality System Documentation
QSD is a paper-based evaluation that serves as a substitute for QMS (formerly GMP).
TFDA issues QMS certificates for manufacturing sites in Taiwan and QSD certificates for manufacturing sites outside of Taiwan.
Other than a handful of low risk, non-sterile medical devices, QSD is a prerequisite for most medical device license. QSD and license applications can be submitted and reviewed in parallel, however, QSD must be approved before TFDA will approve the license application.
QSD must be renewed every 3 years. Applying for QSD renewal is similar to applying for a new QSD in both cost and duration, as it is a substitute for on-site audits.
TFDA issues QMS certificates for manufacturing sites in Taiwan and QSD certificates for manufacturing sites outside of Taiwan.
Other than a handful of low risk, non-sterile medical devices, QSD is a prerequisite for most medical device license. QSD and license applications can be submitted and reviewed in parallel, however, QSD must be approved before TFDA will approve the license application.
QSD must be renewed every 3 years. Applying for QSD renewal is similar to applying for a new QSD in both cost and duration, as it is a substitute for on-site audits.
Required Documents for QSD
Application Type |
Standard |
US Abbreviate |
EU Technical Cooperation Program |
Japan Abbreviate |
Essential |
1. Application forms 2. Medical device distributor certificate 3. General information 4. ISO 13485 certificate 5. Product manufacturing flowchart 6. DMR list |
V |
V |
V |
V |
V |
Plant layout diagram |
V |
V |
V |
||
Layout of production area for each product item |
V |
V |
V |
||
Quality manual |
V |
V |
V |
||
Document master list |
V |
V |
V |
||
Quality SOP |
V |
V (Requires fewer SOP than standard mode) |
|||
CFG |
V |
||||
EIR audit report (or MDSAP report in certain cases) |
V |
||||
FSC |
V |
||||
Latest inspection report issued by the EU NB partners |
V |
||||
On-site inspection reports issued by PMDA or RCBs |
V |
||||
Device Master Records List |
V |
V |
V |
V |
V |
US abbreviate/EU TCP/ Japan abbreviate are available to manufacturing sites located in those regions and meet the abbreviate requirements only.
The EU technical cooperation program includes manufacturing plants in Switzerland and Liechtenstein.
Essential Mode is limited to the scopes mentioned in the annex of article 78 of Medical Device Quality Management System Regulations.
The EU technical cooperation program includes manufacturing plants in Switzerland and Liechtenstein.
Essential Mode is limited to the scopes mentioned in the annex of article 78 of Medical Device Quality Management System Regulations.
Medical Device License
Medical devices are categorized into three classes in accordance to their risk levels. Devices without a previously approved similar product in Taiwan are considered new medical devices and will be subjected to a prolonged reviewing process.
Class 1 |
Class 2 |
Class 3 |
New Medical Device |
(Low Risk) |
(Medium Risk) |
(High Risk) |
(No previously approved Similar Product in Taiwan) |
Good Distribution Practice
According to Article 24 of the Medical Devices Act, in order to ensure that the quality of medical devices can be maintained during the process of storage, transportation, and sales, Medical Device Good Distribution Practice (GDP) is implemented to strengthen the supply chain management of medical devices.
Since May 1, 2023, medical device dealers who hold licenses for 45 scopes of medical devices, and their authorized importers shall establish a system that complies with GDP. Dealers may only wholesale, import, or export after passing the inspection of the central competent authority and obtaining the GDP approval.
Violation of regulations will result in a fine ranging from NT$30,000 to NT$1,000,000 in accordance with Article 70 of the Medical Devices Act.
Since May 1, 2023, medical device dealers who hold licenses for 45 scopes of medical devices, and their authorized importers shall establish a system that complies with GDP. Dealers may only wholesale, import, or export after passing the inspection of the central competent authority and obtaining the GDP approval.
Violation of regulations will result in a fine ranging from NT$30,000 to NT$1,000,000 in accordance with Article 70 of the Medical Devices Act.
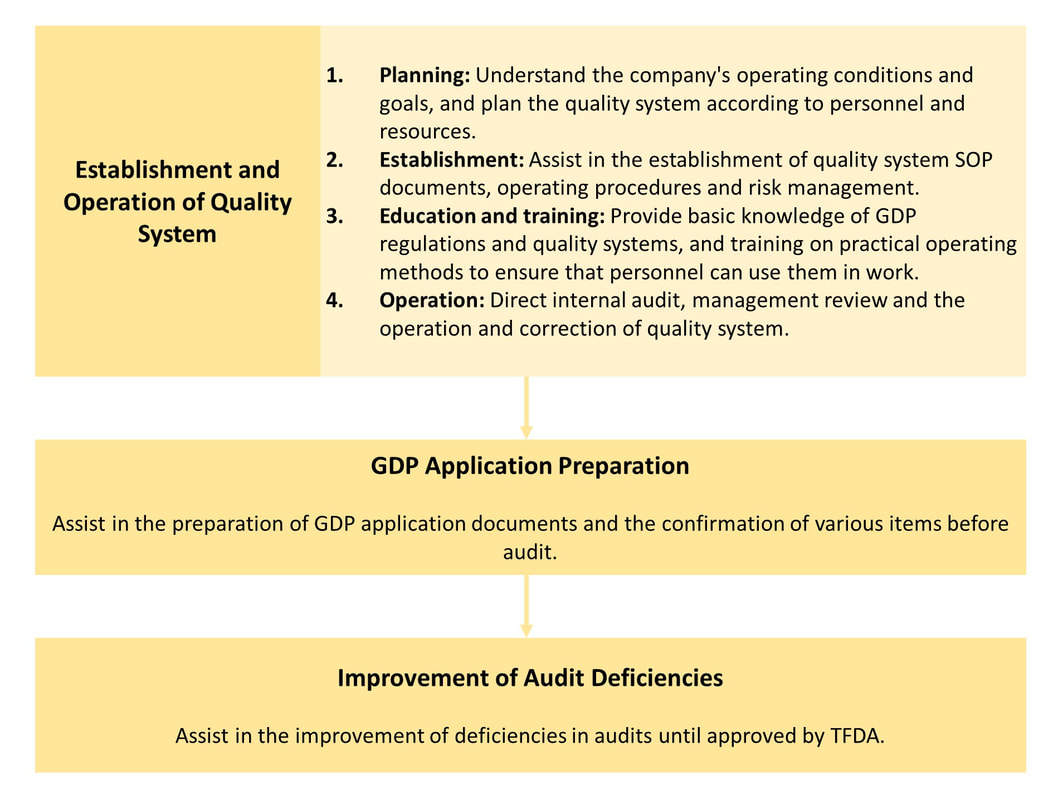
GDP Application |
|
Application Process |
|
Required Documents for Application |
|
Application Fee |
NT$30,000 (One warehouse included. For each additional warehouse that requires on-site inspection, an additional fee of NT$10,000 will be charged. The warehouses that require on-site inspection will be determined by TFDA.) |
Validity Period |
The validity period of GDP approval is 3 years. Those who need a renewal shall apply between 6 to 12 months before the expiry date. |
References |
|
What can we do for you?
Wonderville has obtained TFDA GDP certification and have sufficient knowledge and experience to help you build a quality system complies with GDP.
Commonly asked question
1. Q: What is the difference between listing and licensing for Class I devices?
A: Starting Oct 2021, certain scopes will be registered via listing instead of licensing. Listing will need to be updated annually. Licenses will need to be renewed once every five years.
2. Q: Do I need to provide clinical reports for my product registration?
A: Clinical reports are required for new medical devices. Unless announced otherwise by the TFDA, clinical studies may be waived for class II new medical devices if the following criteria are met:
(a) Product efficacy is not expected to be affected by ethnicity
(b) The intended use or indication of such products has not been reported with serious adverse reactions and has not been required to be removed from any market(s).
(c) The difference between the product and the approved products in Taiwan may be confirmed by pre-clinical data (including tests) to not affect the safety and effectiveness of the product; or the product has been approved/cleared in the United States and the European Union, and the applied indication does not exceed the scope approved by the United States and the European Union.
3. Q: Is there additional rule for my medical device if it is manufactured in China?
A:If your product is manufactured in China, you must confirm that it is not within the restricted items of the International Trade Bureau of the Ministry of Economic Affairs before starting the application.
4. Q: Is there any qualification to apply for medical device license/listing in Taiwan?
A: Prerequisites to medical device licenses:
(a) licenses can only be held by companies with medical device distributor certificates. The certificate must include the scope “import” and at least one technician must be listed. You can consult your accountant in Taiwan for company set up.
(b) Except certain low risk, non-sterile medical device scopes, the manufacturing site’s QSD must be approved before the license can be granted.
5. Q: I am a license holder but I have a distributor in Taiwan, can a different company use my license?
A: The license holder can authorize other medical device distributors to import medical devices directly. For example, if company A has a license and company B wants to be the distributor, both company A and B need to apply for “import authorization” at TFDA. After TFDA approves the import authorization, both companies can use the license to import products into Taiwan.
6. Q: I would like to import a Chinese made medical device. It is approved by CFDA. What do I need to provide?
A: You will need:
(a) A medical device distributor certificate with the“import” scope. Your company must have registered “technician” at the local department of health before you can include “import” on the certificate.
(b) QSD
(c) A Free sale certificate issued by the Chinese health authority clearly stating your product is approved as a medical device (medical equipment) in China, and it must be legalized.
(d) Check the CCC code with the Department of customs. You can check the code at https://fbfh.trade.gov.tw/fh/ap/queryMLFormf.do before filing an official request with department of customs.
(e) Product registration: FSC, LOA, QSD, preclinical test reports (such as functional tests, biocompatibility tests, sterilization validation, software v&v), product information (such as engineering drawings, materials, product specs), final QC … etc.
7. Q: Do I need a GDP certificate?
A: There are 45 scopes of medical devices that will require GDP starting May 2023, you may find the list here https://www.fda.gov.tw/TC/newsContent.aspx?cid=3&id=26852
A: Starting Oct 2021, certain scopes will be registered via listing instead of licensing. Listing will need to be updated annually. Licenses will need to be renewed once every five years.
2. Q: Do I need to provide clinical reports for my product registration?
A: Clinical reports are required for new medical devices. Unless announced otherwise by the TFDA, clinical studies may be waived for class II new medical devices if the following criteria are met:
(a) Product efficacy is not expected to be affected by ethnicity
(b) The intended use or indication of such products has not been reported with serious adverse reactions and has not been required to be removed from any market(s).
(c) The difference between the product and the approved products in Taiwan may be confirmed by pre-clinical data (including tests) to not affect the safety and effectiveness of the product; or the product has been approved/cleared in the United States and the European Union, and the applied indication does not exceed the scope approved by the United States and the European Union.
3. Q: Is there additional rule for my medical device if it is manufactured in China?
A:If your product is manufactured in China, you must confirm that it is not within the restricted items of the International Trade Bureau of the Ministry of Economic Affairs before starting the application.
4. Q: Is there any qualification to apply for medical device license/listing in Taiwan?
A: Prerequisites to medical device licenses:
(a) licenses can only be held by companies with medical device distributor certificates. The certificate must include the scope “import” and at least one technician must be listed. You can consult your accountant in Taiwan for company set up.
(b) Except certain low risk, non-sterile medical device scopes, the manufacturing site’s QSD must be approved before the license can be granted.
5. Q: I am a license holder but I have a distributor in Taiwan, can a different company use my license?
A: The license holder can authorize other medical device distributors to import medical devices directly. For example, if company A has a license and company B wants to be the distributor, both company A and B need to apply for “import authorization” at TFDA. After TFDA approves the import authorization, both companies can use the license to import products into Taiwan.
6. Q: I would like to import a Chinese made medical device. It is approved by CFDA. What do I need to provide?
A: You will need:
(a) A medical device distributor certificate with the“import” scope. Your company must have registered “technician” at the local department of health before you can include “import” on the certificate.
(b) QSD
(c) A Free sale certificate issued by the Chinese health authority clearly stating your product is approved as a medical device (medical equipment) in China, and it must be legalized.
(d) Check the CCC code with the Department of customs. You can check the code at https://fbfh.trade.gov.tw/fh/ap/queryMLFormf.do before filing an official request with department of customs.
(e) Product registration: FSC, LOA, QSD, preclinical test reports (such as functional tests, biocompatibility tests, sterilization validation, software v&v), product information (such as engineering drawings, materials, product specs), final QC … etc.
7. Q: Do I need a GDP certificate?
A: There are 45 scopes of medical devices that will require GDP starting May 2023, you may find the list here https://www.fda.gov.tw/TC/newsContent.aspx?cid=3&id=26852